What Are Good Manufacturing Practices (GMP)?
Good manufacturing practices (GMP) are a system of procedures and controls used to ensure products are consistently produced and meet defined quality standards. It is commonly used in pharmaceuticals, food production and medical device manufacturing to prevent contamination, errors and deviations during production. GMP establishes documented processes, trained personnel requirements and controlled environments to maintain product safety and reliability.
ProjectManager is an award-winning project management software that is equipped with tools that allow manufacturers to plan, schedule and monitor their projects from start to finish. Use Gantt charts to make production roadmaps, manage workflows with kanban boards, allocate resources with workload charts, track costs with timesheets and monitor your projects with real-time dashboards and reports. Get started for free today.

Why Are Good Manufacturing Practices Important?
Manufacturers operate in environments where small process failures can lead to costly defects, safety risks or regulatory action. Implementing good manufacturing practices creates structure across operations, ensuring work is performed consistently, documented properly and monitored continuously. As production planning scales, this level of control becomes essential to maintain quality, meet compliance requirements and avoid disruptions that impact delivery, reputation and profitability.
- Passing audits and inspections required for certifications or licenses
- Ensuring traceability when investigating defects or conducting product recalls
- Maintaining consistent product quality across multiple manufacturing projects
- Supporting global distribution by complying with international GMP standards
- Avoiding production shutdowns caused by compliance violations
- Managing supplier quality and ensuring incoming materials meet specifications
- Protecting brand reputation when operating in safety-critical industries
Benefits of Implementing Good Manufacturing Practices
Strong operational discipline creates measurable advantages beyond compliance. Adopting good manufacturing practices helps manufacturers standardize workflows, reduce variability and improve visibility across production. Over time, this leads to fewer defects, better resource utilization and more predictable outcomes. As teams follow structured processes and maintain accurate records, decision-making becomes faster, risks are easier to manage and overall operational performance improves.
- Improves product quality by enforcing standardized production processes
- Reduces rework and scrap caused by process inconsistencies
- Minimizes downtime through better maintenance planning and process control
- Enhances workplace safety by enforcing operational standards
- Strengthens traceability across materials, batches and finished goods
- Reduces compliance risks and potential regulatory penalties
- Improves supplier coordination and material quality control
20 Good Manufacturing Practices (GMP)
Rather than a single rule, good manufacturing practices (GMP) are applied through a set of operational controls that govern how work is performed, monitored and improved. Each area focuses on a specific part of the production system, helping manufacturers maintain consistency, reduce risk and meet compliance requirements as operations scale.
1. Quality Control (QC)
Quality control is the GMP that defines how materials and products are tested and verified against specifications. It is commonly used in manufacturing to ensure products meet quality standards before release. This includes sampling, testing procedures and laboratory data management.


Testing provides the final confirmation that products meet required standards before reaching customers. Without effective quality control, defects may go unnoticed until after distribution. Strong laboratory controls improve accuracy, support decision-making and ensure only compliant products are released, reducing risk and protecting both the manufacturer and the end user.
2. Facilities & Premises
Facilities and premises is a GMP area that defines how manufacturing spaces are designed, maintained and controlled to support safe production. It is commonly used in regulated environments to prevent contamination, ensure proper workflow and maintain suitable conditions for manufacturing activities. This includes layout design, environmental controls and building maintenance standards. Manufacturers that want to improve the safety, efficiency and productivity of their facilities can try methods such as the gemba walk or a 5s audit.


Production environments directly influence product quality, especially when contamination or cross-contact is a concern. Poor layout design can create bottlenecks or allow materials and personnel to interfere with each other. By maintaining controlled, well-organized facilities, manufacturers reduce operational risks, improve efficiency and ensure that processes can be executed without compromising safety or compliance requirements.
3. Equipment & Utilities
This is one of the most important of all good manufacturing practices, as it governs the selection, operation and maintenance of machinery and support systems used in manufacturing. It is commonly used to ensure equipment performs reliably and utilities like water, air and power meet required standards. This includes calibration, maintenance and performance monitoring procedures.


Reliable equipment and stable utilities are essential for consistent production outcomes. When machines operate outside of specifications or utilities fluctuate, product quality can be affected without immediate detection. Implementing controls around equipment and utilities helps prevent unexpected failures, reduces downtime and ensures that production processes remain stable and repeatable over time.
4. Documentation & Recordkeeping
When it comes to good manufacturing practices (GMP), documentation and recordkeeping establish how procedures, instructions and production activities are documented and stored. It is commonly used in regulated manufacturing to ensure traceability, support audits and verify that processes were followed as intended. This includes standard operating procedures (SOPs), batch records and logs that capture operational data.
Related: 12 Key Manufacturing Documents (with Free Templates)
Clear documentation creates a reliable history of what was planned, executed and verified during production. When records are complete and accurate, teams can trace issues, confirm compliance and support audits without delays. Strong recordkeeping reduces confusion, improves accountability and provides the evidence needed to demonstrate that manufacturing processes are controlled and consistently executed.
5. Materials Management
Materials management controls how raw materials and components are received, stored, handled and tracked throughout the production process. It is commonly used in manufacturing to ensure materials meet specifications and remain suitable for use. This includes supplier verification, inventory tracking and material status identification.
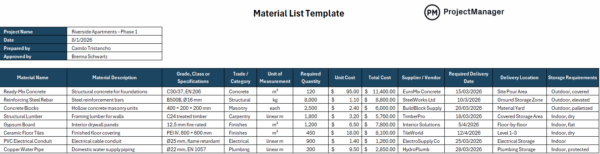

Incoming materials directly affect final product quality, so managing them properly prevents defects from entering production. Without clear controls, expired, contaminated or incorrect materials can be used unintentionally. Establishing structured materials management improves traceability, reduces waste and ensures that only approved inputs are used, supporting consistent and reliable manufacturing outcomes.
6. Production & Process Controls
Production and process controls define how manufacturing operations are executed, monitored and controlled. It is commonly used to ensure processes follow defined parameters and produce consistent results. This includes work instructions, process limits and in-process checks to maintain control during production activities.


Controlled processes reduce variability and make outcomes more predictable across production runs. When operators follow standardized instructions and monitor key parameters, issues can be detected early before affecting larger batches. Strong process controls improve efficiency, minimize rework and help manufacturers maintain consistent product quality while meeting regulatory expectations.
7. Packaging & Labeling
Packaging and labeling is the good manufacturing practices (GMP) area that governs how products are packaged, labeled and prepared for distribution. It is commonly used to ensure correct identification, prevent mix-ups during order fulfillment and provide accurate information to end users. This includes label verification, packaging controls and procedures for handling finished goods.
Incorrect packaging or labeling can lead to serious compliance issues, product recalls or safety risks. By implementing strict controls, manufacturers ensure that the right product is packaged with the correct label every time. This reduces errors, protects consumers and maintains trust while ensuring products meet regulatory and market requirements.
8. Validation & Qualification
Validation and qualification ensure processes, systems and equipment perform consistently as intended. It is commonly used in regulated manufacturing to confirm that production methods produce reliable and repeatable results. This includes process validation, equipment qualification and system verification activities.
Unverified processes can introduce hidden risks that affect product quality over time. By validating systems and qualifying equipment, manufacturers gain confidence that operations are stable and controlled. This reduces uncertainty, prevents recurring issues and supports consistent production performance, especially when scaling operations or introducing new processes or technologies.
9. Maintenance & Calibration
Maintenance and calibration is the process that ensures equipment is serviced and adjusted to operate within defined performance limits. It is commonly used in manufacturing to prevent equipment failures and maintain measurement accuracy. This includes preventive maintenance schedules, calibration procedures and service records to keep systems reliable and compliant.
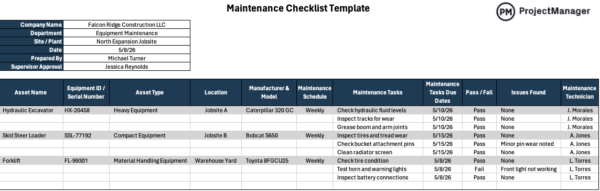

This is one of the most necessary good manufacturing practices of them all as every piece of production equipment tends to drift over time, especially under continuous use in production environments. Without regular maintenance and calibration, machines can produce inconsistent outputs or inaccurate measurements that impact product quality. Establishing structured schedules reduces unexpected downtime, improves reliability and ensures production processes remain stable and within acceptable operating conditions.
10. Sanitation & Hygiene
Sanitation and hygiene is the series of guidelines, cleaning procedures and personal cleanliness standards required to prevent contamination during manufacturing. It is commonly used in food, pharmaceutical and cosmetic production to protect product integrity. This includes cleaning procedures, sanitation schedules and hygiene requirements for personnel and work areas.
Contamination risks increase quickly when cleaning routines are inconsistent or poorly enforced. Even small lapses in hygiene can affect entire production batches, leading to waste or safety concerns. By implementing strict sanitation controls, manufacturers maintain clean environments, reduce contamination risks and ensure products are produced under conditions that support safety and compliance.
11. Environmental Monitoring
Environmental monitoring is the process of tracking conditions such as temperature, humidity, air quality and microbial levels in manufacturing environments. It is commonly used in controlled production settings to ensure environmental factors remain within acceptable limits. This includes routine measurements, testing protocols and recorded data to verify environmental stability.
Production environments can shift due to external factors or equipment changes, affecting product quality without immediate visibility. Monitoring these conditions allows teams to detect deviations early and take corrective action before issues escalate. Consistent environmental tracking supports stable operations, protects sensitive processes and ensures manufacturing conditions remain suitable for producing compliant products.
12. Deviations, CAPA & Change Control
Deviations, CAPA and change control is a GMP area that manages unexpected issues, corrective actions and controlled modifications to processes or systems. It is commonly used to investigate problems, address root causes and implement approved changes without disrupting compliance. This includes deviation reports, corrective action plans and formal change approval workflows.
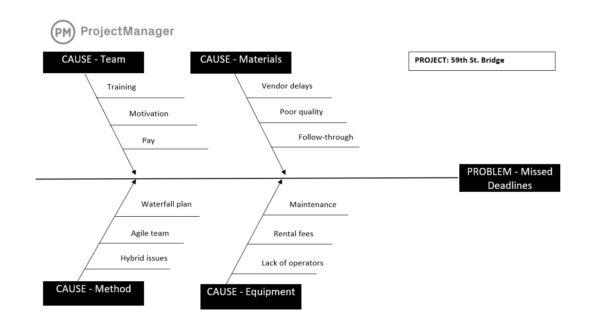

Problems are inevitable in any production environment, but how they are handled determines long-term performance. Without structured controls, the same issues can repeat or changes can introduce new risks. Managing deviations and changes systematically helps resolve issues effectively, prevents recurrence and ensures all modifications are reviewed, approved and documented before implementation.
13. Product Recall & Returns
Product recall and returns are all of the good manufacturing practices that establish how defective or non-compliant products are identified, retrieved and managed after distribution. It is commonly used in manufacturing to remove unsafe products from the market and trace affected batches. This includes recall procedures, return handling and traceability systems to control product movement.
Once products reach customers, any quality issue becomes harder to control without a structured recall process. Delays or gaps in traceability can increase risk and regulatory exposure. Establishing clear recall and return procedures enables fast action, limits impact, protects consumers and helps manufacturers respond effectively when product issues are identified in the market.
14. Supplier Relationship Management
Supplier relationship management governs how external partners are selected and monitored to ensure consistent input quality. It is commonly used in manufacturing to verify supplier reliability and maintain material standards. This includes supplier qualification, performance evaluation and approved vendor lists.
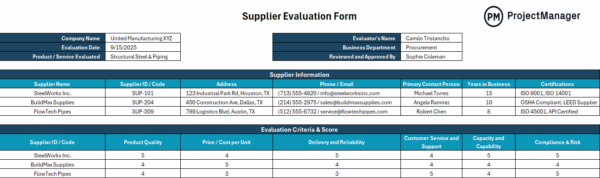

Manufacturing output depends heavily on the quality of incoming materials and outsourced services. Weak supplier oversight can introduce variability or defects before production even begins. By managing vendors through structured evaluations and ongoing monitoring, manufacturers reduce risk, improve consistency and ensure that all inputs align with required specifications and compliance expectations.
15. Distribution & Storage
Distribution and storage is a GMP area that controls how finished products are stored, handled and transported to maintain quality after production. It is commonly used in manufacturing to ensure products remain within defined conditions until they reach customers. This includes storage controls, inventory management and transportation requirements.
Conditions during storage and distribution can affect product integrity, especially for temperature-sensitive or regulated goods. Poor handling or inadequate storage can lead to degradation, contamination or non-compliance. By establishing clear controls over storage environments and transportation processes, manufacturers protect product quality, reduce losses and ensure products reach customers in acceptable condition.
How ProjectManager Helps Manufacturing Businesses
Keeping production on schedule while managing resources, costs and changing priorities requires more than spreadsheets or disconnected tools. Manufacturing project management becomes easier when teams can plan, track and adjust work in one centralized system. ProjectManager brings together production scheduling, resource planning and performance tracking so production teams can stay aligned and make faster decisions on the shop floor.
With interactive Gantt charts, manufacturing teams can build detailed production schedules, map task dependencies, identify milestones and adjust timelines as conditions change. Resource management tools allow managers to assign labor, equipment and materials efficiently, while monitoring resource availability and avoiding bottlenecks. Real-time dashboards and reports provide instant visibility into progress, costs and workload, helping teams identify issues early and maintain control over production performance.
Teams can also collaborate more effectively using kanban boards and task lists that track work at every stage of the manufacturing process. Thanks to these and other tools and features, ProjectManager helps manufacturing businesses improve efficiency, reduce delays and deliver consistent results. Watch the video below to learn more!
ProjectManager is award-winning software for managing any project. Our collaborative platform connects your teams and gives you access to the manufacturing floor no matter where you are or what time it is. Get started with ProjectManager today for free.